Please see Chapter 4 Chemical Kinetics Exam Questions Class 12 Chemistry below. These important questions with solutions have been prepared based on the latest examination guidelines and syllabus issued by CBSE, NCERT, and KVS. We have provided Class 12 Chemistry Questions and answers for all chapters in your NCERT Book for Class 12 Chemistry. These solved problems for Chemical Kinetics in Class 12 Chemistry will help you to score more marks in upcoming examinations.
Exam Questions Chapter 4 Chemical Kinetics Class 12 Chemistry
Short Answer Type Questions-I
Question. (i) What is the order of the reaction whose rate constant has same units as the rate of reaction ?
(ii) For a reaction A + H2O → B; Rate ∝ [A]. What is the order of this reaction?
Answer. (i) Zero order
(ii) Pseudo-first order
Question. For a reaction the rate law expression is represented as follows:
Rate = k [A][B]1/2
(i) Interpret whether the reaction is elementary or complex. Give reason to support your answer.
(ii) Write the unit of rate constant for this reaction if concentration of A and B is expressed in moles/L.
Answer. (i) This is a complex reaction.
Order of reaction is a fraction value. i.e., 1.5.
Molecularity cannot be a fraction. The reaction occurs in steps, so it is a complex reaction where order and molecularity has different values.
(ii) Rate = k[A][B][½]
Unit of Rate constant (k)

Question. For a reaction:
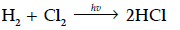
Rate = k
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Answer. (i) Zero order reaction, Molecularity is 2 / bimolecular reaction.
(ii) mol L–1 s–1.
Question. State a condition under which a bimolecular reaction is kinetically first order reaction.
Answer. Let us take a bimolecular reaction :
A + B → Product
Rate = k [A] [B]
When concentration of [B] is taken in excess then rate law will become:
Rate = k [A]
where, k = constant
The rate depends only on one of the reactant as there is negligible change in its concentration so it is bimolecular but is of first order.
Question. (i) For which reaction, the rate of reaction does not decrease with time?
(ii) What is the order of photochemical reaction?
Answer. (i) For zero order reaction the rate of reaction does not decrease with time because it does not depend on concentration of reactants.
(ii) Zero Order reaction.
Question. (i) If the rate equation is given below:
Rate = k[A]2[B]
then what will be the unit of its rate and rate constant?
(ii) Write the slope value obtained in the plot of log [R0] / [R] vs. time for a first order reaction.
Answer.

Question. Write units of rate constants for zero order and for the second order reactions if the concentration is expressed in mol L–1 and time in second.
Answer. Zero order: mol L–1s–1
Second order: L mol–1s–1
Question. For a reaction,
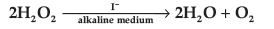
The proposed mechanism is given below:
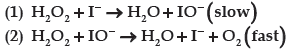
(i) Write rate law of the reaction.
(ii) Write overall order of reaction.
(iii) Out of step (1) and (2), which one is rate determining step?
Answer. (i) Rate = k [H2O2] [ I¯]
(ii) Order = 2
(iii) Step 1
Question. For a chemical reaction R → P, variation in ln [R] vs time (t) plot is given below:

For this reaction:
(i) Predict the order of reaction.
(ii) What is the unit of rate constant (k)?
Answer. (i) First order.
(ii) s-1/time-1
Question. (i) Why the molecularity of a reaction can not be zero?
(ii) Write the formula for expressing rate of the reaction.
N2 + 3H2 → 2NH3
Answer. (i) The molecularity of a reaction is the number of total molecules taking part in elementary step of reaction. So, minimum one molecule is required for a reaction to occur. Hence, the value of molecularity is never zero.
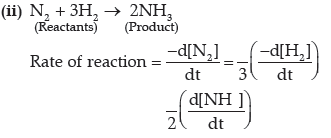
Question. The C-14 content of an ancient piece of wood was found to have three tenths of that in living trees.
How old is that piece of wood? (log 3= 0.4771, log 7 = 0.8540 , Half-life of C-14 = 5730 years)
Answer.

Question. For a chemical reaction R → P, variation in log [Ro]/[R] vs time plot is given below:
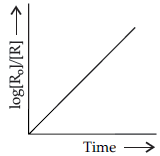
For this reaction:
(i) Predict the order of reaction
(ii) What is the unit of rate constant (k)?
Answer. (i) First order.
(ii) s-1/time-1
Question. Explain the following terms:
(i) Rate constant (k)
(ii) Half-life period of reaction (t1/2).
Answer. (i) Rate constant (k): Rate constant is rate of the reaction when the concentration of reactants is unity.
(ii) Half-life period of a reaction (t1/2): Half-life of a reaction is the time in which the concentration of a reactant is reduced to half of its original value.
Question. (i) Explain why H2 and O2 do not react at room temperature.
(ii) Write the rate equation for the reaction
A2 + 3B2 → 2C, if the overall order of the reaction is zero.
Answer. (i) Due to high activation energy of the reaction between O2 and H2.
(ii) Rate = k [A2]0[B2]0
Question. For a reaction:

Rate = k ;
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Answer. (i) Zero order, bimolecular / unimolecular.
(ii) mol L–1 s–1.
Question. Derive integrated rate equation for rate constant of a first order reaction.
Answer.

Question. For the reaction
2N2O5 (g) → 4NO2 (g) + O2 (g),the rate of formation of NO2 (g) is 2.8 × 10–3 M s–1. Calculate the rate of disappearance of N2O5 (g).
Answer.
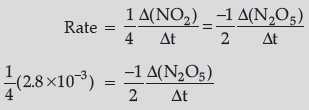
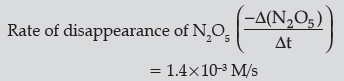
Question. A first-order reaction has a rate constant 1.15 × 10−3 s−1. How long will 5 g of this reactant take to reduce to 3 g ?
Answer. Initial amount = 5 g
Final concentration = 3 g
Rate constant = 1.15 × 10–3 s–1
We know that for a First order reaction,
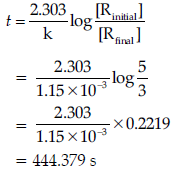
Question. Define order of reaction. Predict the order of reaction in the given graphs:

Where [R]0 is the initial concentration of reactant and t1/2 is half life.
Answer. Order of reaction is defined as the sum of powers to which the concentration terms are raised in the rate law equation.
(a) First order (b) zero order
Question. For a certain chemical reaction variation in concentration [A] vs. time (s) plot is given below:
(i) Predict the order of the given reaction?
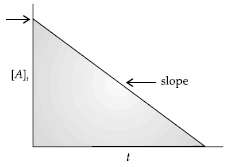
(ii) What does the slope of the line and intercept indicate?
(iii) What is the unit of rate constant k?
Answer. (i) Zero order reaction
(ii) Slope represents –k ; Intercept represents [A]0
(iii) mol L–1 s–1
Question. (i) The rate of reaction decreases with the progress of reaction. Why?
(ii) Reactions having molecularity more than three occur rarely. Why?
Answer. (i) The rate of reaction depends on the concentration of reactants. Since, the concentration of reactants decreases with time, so rate of reaction also decreases.
(ii) It is because of the fact that collision of more than three molecules is not possible at a time.
Question. The following results have been obtained during the kinetic studies for the reaction:
P + 2Q → R + 2S
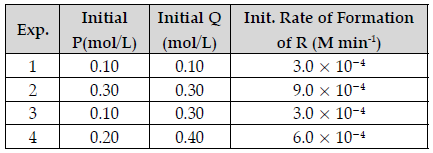
Determine the rate law expression for the reaction.
Answer. Let the rate law expression be Rate = k [P]x [Q]y from the table we know that
Rate 1 = 3.0 × 10–4 = k (0.10)x (0. 10)y
Rate 2 = 9.0 × 10–4 = k (0.30)x (0.30)y
Rate 3 = 3.0 × 10–4 = k (0.10)x (0.30)y
Rate 1/ Rate 3 = (1/3)y or 1 = (1/3)y
So, y = 0
Rate 2/ Rate 3 = (3)x or 3 = (3)y
So, x = 1
Rate = k [P]
Question. (i) What is the order of the reaction whose rate constant has same units as the rate of reaction ?
(ii) For a reaction A + H2O → B; Rate ∝ [A].
What is the order of this reaction ?
Answer. (i) Zero order
(ii) Pseudo-first order
Short Answer Type Questions-II
Question. For the first order thermal decomposition reaction, the following data were obtained:
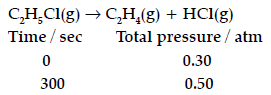
Calculate the rate constant
(Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
Answer. Given: Initial pressure,
P0 = 0.30 atm
Pt = 0.50 atm
t = 300 s
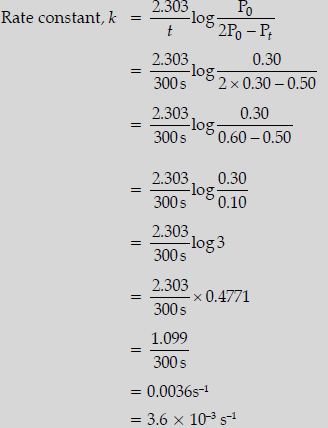
Question. The decomposition of NH3 on platinum surface is a zero order reaction. What will be rate of production of N2 and H2 when the value of k is 2.5 × 10−9 mol L–1 s–1?
Answer.

Question. For the reaction
2A + B → A2B
Rate = k[A] [B]2, k = 2.0 × 10–6 mol–2L2s–1
Calculate the initial rate of the reaction when [A] = 0.1 mol L–1 and [B] = 0.2 mol L–1 . Calculate the rate of reaction after [A] is reduced to 0.06 mol L–1.
Answer. 2A + B → A2B
Initial rate = k[A] [B]2
= 2.0 × 10−6 × 0.1 × (0.2)2
= 8.0 × 10−9 mol L–1
When concentration of [A] is reduced from 0.10 mol L–1 to 0.06 mol L–1 i.e., 0.04 mol L–1,concentration of A has been used in the reaction.
Therefore, the concentration of B
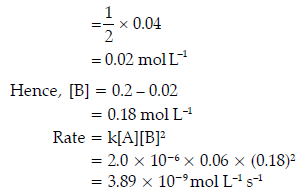
Question. For the reaction R → P, the concentration of a reactant changes from 0.03 to 0.02 M in 25 minutes.
Calculate the average rate of reaction using units of time both in minutes and seconds.
Answer. R → P
[R1] = 0.03 M
[R2] = 0.02 M
Δt = 25 min

Question. Following data are obtained for the reaction:
N2O5 → 2NO2 + ½O2

(a) Show that it follows first order reaction.
(b) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
Answer.
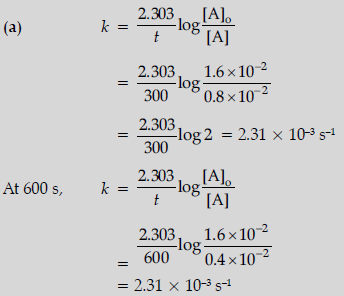
k is constant when using first order equation therefore it follows first order kinetics.
OR
In equal time interval, half of the reactant gets converted into product and the rate of reaction is independent of concentration of reactant, so it is a first order reaction.
(b) t1/2 = 0.693/k
= 0.693/2.31 × 10–3
= 300 s
Question. A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given: log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
Answer.
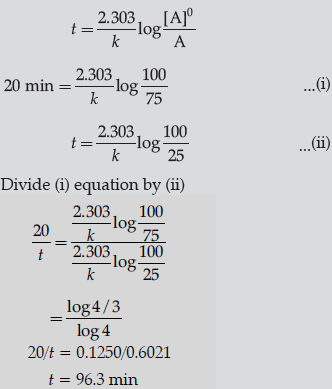
Question. The following data were obtained for the reaction:
A + 2B → C
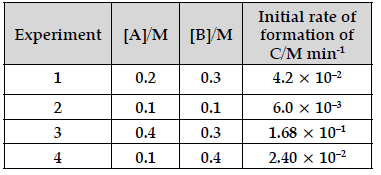
(a) Find the order of reaction with respect to A and B.
(b) Write the rate law and overall order of reaction.
(c) Calculate the rate constant (k).
Answer. Rate = k[A]p[B]q
On solving
(a) Order with respect to A = 2 , B = 1
(b) Rate = k[A]2[B]1 ; overall order = 3
(c) Experiment 1: 4.2 × 10−2 = k(0.2)2 (0.3); k = 3.5
Experiment 2: 6.0 × 10−3 = k(0.1)2 (0.1) ; k = 6
Long Answer Type Questions
Question. (i) Write the rate law for a first order reaction. Justify the statement that half life for a first order reaction is independent of the initial concentration of the reactant.
(ii) For a first order reaction, show that the time required for 99% completion of a first order reaction is twice the time required for the completion of 90%.
Answer. (i) For a first order reaction
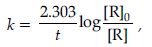
where [R]0 = initial concentration, [R] = conc. after time t
When half of the reaction is completed, [R] = [R]0/2. Representing, the time taken for half of the reaction to be completed, by t1/2, equation becomes:
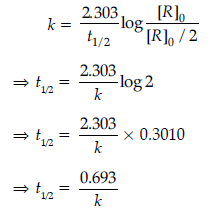
The above equation shows that half-life of first order reaction is independent of the initial concentration of the reactant.
(ii) For a first order reaction

Question. (a) A first order reaction is 25% complete in 40 minutes.
Calculate the value of rate constant. In what time will the reaction be 80% completed?
(b) Define order of reaction. Write the condition under which a bimolecular reaction follows first order kinetics.
Answer.
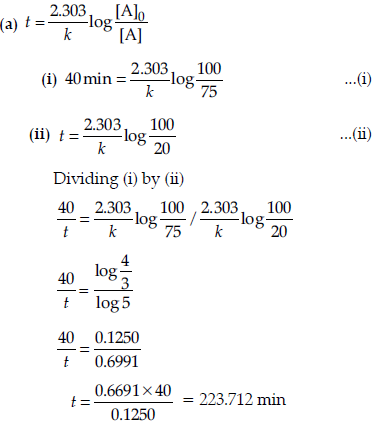
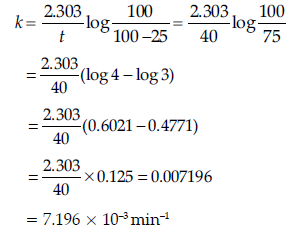
(b) Order of reaction: The sum of the coefficients of the reacting species that are involved in the rate equation for the reaction, is called order of reaction.
The condition under which a bimolecular reaction follows first order kinetics is when one of the reactants is taken in large excess that its concentration hardly changes.
Question. A reaction is of first order with respect to A and second order with respect to B
(i) Write differential rate equation.
(ii) How is the rate effected when the concentration of B is tripled?
(iii) How is the rate affected when the concentration of both A and B is doubled?
Answer. (i) Differential rate equation
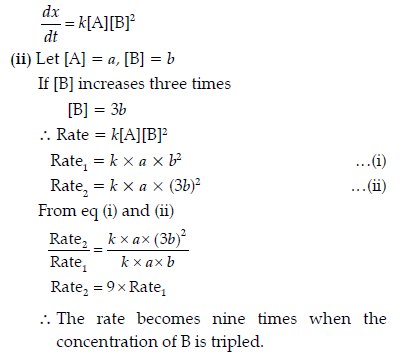
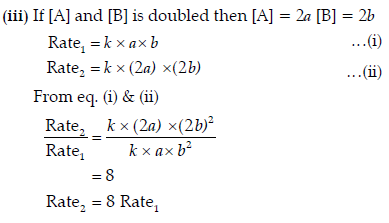
∴ The rate becomes eight times when the concentration of both A and B is doubled.
Question. (i) A first order reaction is 75% completed in 40 minutes. Calculate its t1/2.
(ii) Predict the order of the reaction in the given plots:

where [R]0 is the initial concentration of reactant.
(Given: log 2 = 0.3010, log 4 = 0.6021)
Answer.
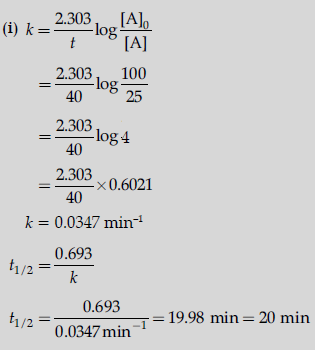
(ii) (a) First order reaction
(b) Zero order reaction
Question. For a given chemical reaction determine the order of reaction with respect to HgCl2, with respect to C2O42– and overall reaction.
HgCl2(aq) + C2O42–(aq) → 2 Cl–(aq) + 2 CO2(g) + Hg2Cl2(s)
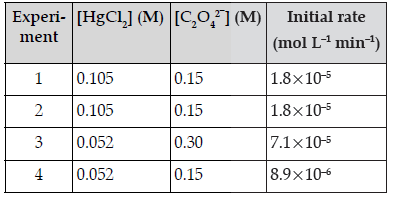
Answer. From the reaction, rate law will be expressed as follows, Rate = k[HgCl2]m[C2O42–]n Compare the rates in experiments 1 and 2 (or 3 and 4) to find the order in oxalate ion:

0.493 = 0.495m
m = 1
Therefore, the reaction is first order with respect to mercury (II) chloride and second order with respect to oxalate. The overall order = m + n = 2 + 1 = 3,third order.
Question. (i) For a reaction A + B → P, the rate is given by Rate = k[A] [B]2
(a) How is the rate of reaction affected if the concentration of B is doubled ?
(b) What is the overall order of reaction if A is present in large excess ?
(ii) A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction. (log 2 = 0.3010) U[Question for Green board]
Answer. (i) A + B → P
Rate = k[A] [B]2
(a) When concentration of B is doubled it means concentration of B becomes 2 times.
Thus, Rate = k[A]1 [2B]2
= k[A] [4B2]
So, the rate becomes 4 times.
(b) Order of reaction is the number of molecules whose concentration alters after the reaction. If A is present in excess i.e., its concentration is unaffected.
So, rate depends only on the concentration of B. as k = [B]2
Thus, the reaction is of second order.
(ii) STEP 1: For the 1st order reaction:
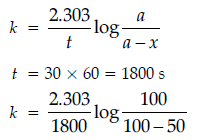

Question. (a) Reaction 2A + B + C → D + 2E shows first order with respect to A, second order with respect to B and zero order with respect to C. Determine:
(i) Rate law of reaction
(ii) What will be the rate of reaction on doubling of concentration of A, B and C?
(b) For the reaction,

The rate of dissociation of N2O5 is 5.65 × 10–5 mol L–1 s–1. Determine:
(i) Rate of reaction
(ii) Rate of formation of NO2
(iii) Rate of formation of O2
Answer. (a) (i) 2A + B + C → D + 2E
Rate Law
Rate (r) = k[A]1 [B]2[C]0
(ii) If concentration of A, B & C is doubled
r1 = k[A]1 [B]2[C]0 …(i)
r2 = k[2A]1 [2B]2[2C]0 …(ii)
From equation (i) & (ii)

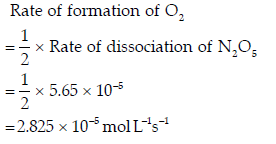
Question. The following data were obtained for the reaction:
2 NO + O2 → 2 NO2
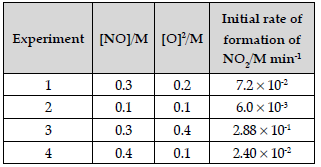
(i) Find the order of reaction with respect to NO and O2.
(ii) Write the rate law and overall order of reaction.
(iii) Calculate the rate constant (k).
Answer. (i) Rate = k[NO]x[O2]y
7.2 × 10-2 = k[0.3]x[0.2]y Eqn (1)
6.0 × 10-3 = k[0.1]x[0.1]y Eqn (2)
2.88 × 10-1 = k[0.3]x[0.4]y Eqn (3)
2.40 × 10-2 = k[0.4]x[0.1]y Eqn (4)

