Please refer to the Carbon and Its Compound Revision Notes given below. These revision notes have been designed as per the latest NCERT, CBSE and KVS books issued for the current academic year. Students will be able to understand the entire chapter in your class 10th Science book. We have provided chapter wise Notes for Class 10 Science as per the latest examination pattern.
Revision Notes Chapter 4 Carbon and Its Compound
Students of Class 10 Science will be able to revise the entire chapter and also learn all important concepts based on the topic wise notes given below. Our best teachers for Grade 10 have prepared these to help you get better marks in upcoming examinations. These revision notes cover all important topics given in this chapter.
The chemical symbol of Carbon is C. Its atomic number is 6 and mass number is 12.It is a nonmetallic element.
ALLOTROPES OF CARBON

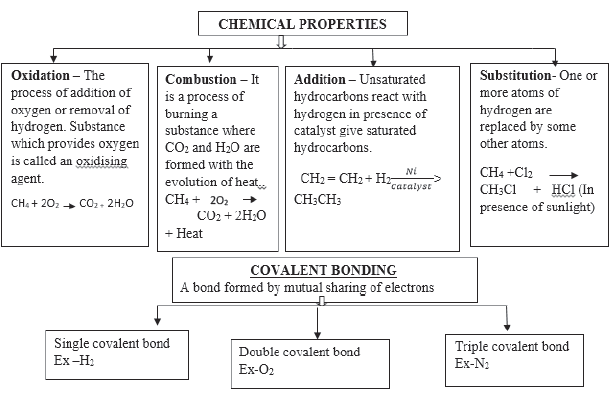
VERSATILE NATURE OF CARBON

ISOMERISM: The phenomenon in which a compound has the same molecular formula but different structures is called isomerism.
ISOMERS: The compounds which have the same molecular formula but different structures and different properties are called isomers.
FUNCTIONAL GROUPS: Atoms or group of atoms responsible for the chemical properties of an organic compound.

In a hydrocarbon chain, one or more hydrogens can be replaced by some other element . In such compounds, the element replacing hydrogen is referred to as a heteroatom

HOMOLOGOUS SERIES: A series of compounds having same functional group and similar chemical properties but differ by–CH2 unit between two successive members.