Students should practice questions given in Is Matter Around Us Pure Chapter 2 Class 9 Science Worksheets. These worksheets for Class 9 Science have a good collection of important questions and answers which are expected to come in your class tests and examinations. You should learn these solved worksheet questions for Science Class 9 as it will help you to understand all topics and give you more marks.
Class 9 Science Worksheets Chapter 2 Is Matter Around Us Pure
Please refer to below questions and answers for Is Matter Around Us Pure Chapter 2 Class 9 Science Worksheets. Prepared by expert teachers for Standard 9 Science
Question. What is the meaning of ‘Kroma’?
Answer
‘Kroma’ means colour in Greek.
Question. Naphthalene and sand can be separated by the process of-
Answer
Sublimation.
Question. Define the term Solvent.
Answer
Component of solution that is present in the larger proportion and dissolves the other substance is called Solvent.
Question. Name the constituents of German silver.
Answer
Copper and zinc.
Question. Identify the following as physical or chemical changes :
(a) Formation of cloud
(b) Magnetizing a iron nail
(c) Water boils to form steam
(d) An almirah gets rusted
Answer
(a) Formation of cloud Physical change
(b) Magnetizing a iron nail Physical change
(c) Water boils to form steam Physical change
(d) An almirah gets rusted Chemical change
Question. A homogeneous mixture of two or more substances called-
Answer
Solution.
Question. A colloid which have liquid as dispersed phase and solid as dispersion medium called-
Answer
Gel.
Question. Name two metals which are highly malleable and ductile.
Answer
Gold and silver.
Question. The component of solution that is present in smaller proportion
Answer
Solute.
Question. What are two types of matter on the basis of composition?
Answer
Pure substance and mixture.
Question. Give natural example of mixture.
Answer
Sea water, minerals, soil.
Question. Name two categories of pure substance.
Answer
Element and compound.
Question. Write the name of any two compounds which sublime on heating.
Answer
Ammonium chloride and iodine.
Question. Name the process by which pure copper sulphate can be obtained from its impure sample.
Answer
Crystallization.
Question. Give two examples of colloids from your daily life.
Answer
Milk and fog.
Question. Name the process by which all dyes present in black ink be recovered.
Answer
Chromatography.
Question36. Draw a flow chart showing the separation of components of Air.
Answer. Air is a homogeneous mixture and its components can be separated by fractional distillation.
Steps : Flow chart :

Question. Give difference between mixture and compound.
Answer.
Compounds Mixtures
1. Compounds are pure Mixtures are impure substances.
substances.
2. Compounds are made Mixtures are made up of two or more substances
up of two or more mixed physically.
elements combined
chemically.
3. The components of a The components of a mixture are
compound are present present in different ratio.
in a fixed ratio.
4. Compounds have Mixtures do not have same properties
same properties throughout the mixture part.
throughout the
compound part.
5. A new substance No new substance is formed.
is formed.
6. The components of The components of a mixture can be
a compound can be separated by physical methods.
separated only by
chemical methods.
Question. Write the properties of a Solution.
Answer.
(i) It is a homogenous mixture of solute and solvent.
(ii) Solute particles cannot be separated by filtration.
(iii) True solution is clear and transparent.
(iv) True solution does not scatter light.
(v) Solute particles cannot settle out from solvent.
Question. Why is water considered as compound?
Answer. Water is considered as compound because :
(i) Water is composed of two elements : hydrogen and oxygen.
(ii) The ratio of hydrogen and oxygen by mass in any sample of pure water is the same.
(iii) The properties of water are different from its constituent elements : hydrogen and oxygen.
(iv) Water can be decomposed by chemical means only (e.g. electrolytically) into hydrogen and oxygen.
Question. What makes water as a universal solvent?
Answer. Water acts as a universal solution due to :
(i) The polar nature of its molecules.
(ii) Its ability to produce soluble salt on interaction with a large number of substances.
Question. Melting of wax is a physical change but burning of wax is a chemical change. Explain.
Answer.
(i) Melting of wax changes the state of wax from solid to liquid. No new substance is formed. The chemical nature of solid wax and liquid wax is the same. Hence, melting of wax is a physical change.
(ii) Burning of wax gives new gaseous products. Both the physical and chemical changes are taking place. Hence, burning of wax is a chemical change.
Question. (i) State the principle of the process of centrifugation.
(ii) List any three applications of centrifugation.
Answer.
(i) Centrifugation is the process of separating suspended particles from a liquid like colloids by churning the liquid at a high speed. The principle is that denser particles are forced to the bottom and lighter stay at the top when spun rapidly.
(ii) Applications of centrifugation :
(a) Used in dairies and homes to separate cream from milk or butter from cream.
(b) Used in washing machines to squeeze out water from clothes.
(c) Used in laboratories to separate colloidal particles from their solutions.
(d) Used in diagnostic labs for blood and urine test.
Question. Calculate the grams of NaC1 (5.25% by mass) in 245 grams of a commercial bleach solution.
Answer.
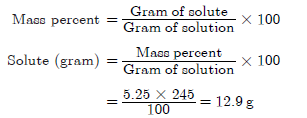
Question. Give difference between true solutions and colloidal solutions.
Answer.

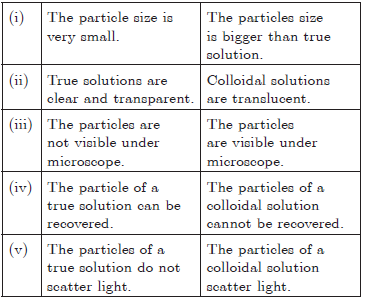
Question. Explain sublimation process with labelled diagram.
Answer. The process of converting solid form directly into vapour form is called sublimation. This is a reversible process.
Process :
(i) Take some camphor or ammonium chloride.
(ii) Crush the compound and put it in china dish.
(iii) Fit an inverted funnel over the china dish.
(iv) Put a cotton ball on the stem of the funnel.
(v) Heat the china dish slowly and observe.
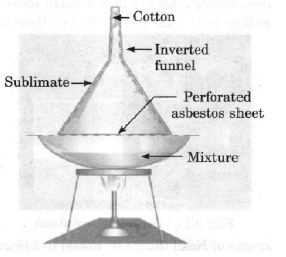
Figure: Separation of ammonium chloride and salt by sublimation Ammonium chloride is the molecule which on heating changes directly into gaseous state. When vapours of ammonium chloride collected then solid ammonium chloride is formed.
NH4Cl (solid) + NH4Cl (vapour)
Observation : A change of state directly from solid to gas without changing into liquid state (or vice versa) is observed, this phenomenon is called sublimation.
Question. What are colloids? Give their characteristics.
Answer. Colloidal solution is a heterogeneous mixture whose particles are bigger than size of particle in solution but cannot be visible by naked eye. When the beam of light passes through a colloid, then the path of light becomes visible. For example : Milk, smoke, etc.
(i) Brownian movement : The particles of a colloid show Brownian movement. The particles in it never fully settle down even leaving for a long time to remain undisturbed. They show constant zigzag movement. The type of movement is caused by the constant collisions between the particles of the dispersing medium and dispersed phase.
(ii) Tyndall effect : If the beam of light passes through a colloid then the path of light becomes visible. This is called Tyndall effect. The colloidal particles shine because they scatter the light falling on them in all directions.
