Please see Chapter 10 Metals and Non-Metals Exam Questions Class 10 Science below. These important questions with solutions have been prepared based on the latest examination guidelines and syllabus issued by CBSE, NCERT, and KVS. We have provided Class 10 Science Questions and answers for all chapters in your NCERT Book for Class 10 Science. These solved problems for Metals and Non-Metals in Class 10 Science will help you to score more marks in upcoming examinations.
Exam Questions Chapter 10 Metals and Non-Metals Class 10 Science
ONE MARK QUESTIONS
Question: Give one most suitable word for the following statements:
a. Metal oxides which show basic as well as acidic behaviour.
b. Iodine, a shining non-metal.
Answer: a. Amphoteric oxides.
b. Lustrous.
Question: Why oxides of highly reactive metals cannot be reduced by carbon?
Answer: It is because highly reactive metals themselves are good reducing agents, so they can’t be reduced by carbon.
Question: Give reason why: Electric wires are coated with plastic.
Answer: Plastic is a non-conductor of electricity, therefore electric wires are coated with plastic.
Question: Give composition of the alloy: brass and bronze.
Answer: Brass is made up of Cu and Zn. Bronze is made up of Cu and Sn.
Question: Write the chemical equation for the reaction taking place when steam is passed over hot Aluminium?
Answer: 2Al(s) + 3H2O(aq) Heat→ Al2O3(S) + 3H2(g)
Question: An alloy has low melting point and is therefore used for electrical fuse. Name the alloy and write its constituents.
Answer: Solder is an alloy. It is made up lead and tin.
Question: What happens when carbon dioxide is compressed in water at high pressure?
Answer: CO2 + H2O → H2CO3
Carbonic acid is formed.
Question: Name two metals which react with dilute HNO3 to evolve H2 gas.
Answer: Mn and Mg.
Question: What is the formula of rust?
Answer: Rust is hydrated ferric oxide, Fe2O3 ·xH2O .
Question: Why sulphide and carbonate ores are converted into oxides?
Answer: It is because oxides are more easier to be reduced than sulphides and carbonates.
Question: Name one metal and one non-metal in liquid state at room temperature.
Answer: Mercury is a metal and Bromine is a non-metal present as a liquid at room temperature.
Question: From amongst the metal, sodium, calcium, aluminium, copper and magnesium, name the metal:
a. which reacts with water only on boiling.
b. another one which does not react even with steam.
Answer: a. Mg, b. Cu
Question: What is formed when sodium absorb moisture from air? Give equation also.
Answer: Sodium hydroxide and hydrogen gas is formed:
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
Question: Name any one metal which reacts neither with cold water nor with hot water but reacts with heated steam to produce hydrogen gas.
Answer: Iron (Fe) :
3Fe(s) + 4H2O(g) Heat→ Fe3O4(s) + 4H2(g)
Question: A green layer is gradually formed on copper plate when left exposed to air for a week in a bathroom. What could this green substance be?
Answer: CuCO3.Cu(OH)2, Basic copper carbonate.
Question: Name the metals Which have low melting point.
Answer: Gallium and Caesium.
Question: Define metallurgy.
Answer: Metallurgy. All the processes involved in the extraction
of metals from its ore is called metallurgy.
Question: Choose the amphoteric oxide amongst the following:
Na2O, ZnO, Al2O3, CO2, H2O
Answer: ZnO and Al2O3 are amphoteric oxides.
Question:Name a non-metal which is lustrous whereas a metal which is non-lustrous.
Answer: Iodine is a lustrous non-metal, Lead is a non-lustrous metal.
Question: What kind of compounds are called Ionic compounds?
Answer: Those compounds which are formed between metals and non-metals by transfer of electrons e.g., NaCl, KCl etc.
Question: Why do ionic compounds have high melting point?
Answer: It is due to strong forces of attraction between positively charged and negatively charged ions.
Question: Write a balanced chemical equation for the reaction:
Aluminium when heated in air. Write the name of the product formed.
Answer: 4Al(s) + 3O2(g) Heat→ 2Al2O3(S)
Aluminium oxide
TWO MARKS QUESTIONS
Question: (a) In electrolytic refining of impure copper metal, what are used as cathode and anode?
(b) Show the formation of HgCl2 from magnesium and chlorine atoms.
Answer: (a) Pure copper is used as the cathode, impure copper is used as the anode.
(b)
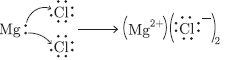
Question: Name a metal/non-metal
a. Which makes iron hard and strong?
b. Which is alloyed with other metal to make amalgam?
c. Which is used to galvanise iron articles?
d. Whose articles when exposed to air forms a black coating?
Answer: a. Carbon makes it hard and strong. Tungsten also makes it hard and strong.
b. Mercury is alloyed with metals to form amalgam.
c. Zinc metal is used for coating over iron metal to galvanise iron.
d. Silver articles acquire black coating when left exposed to the atmosphere.
Question: The reaction of metal X with Fe2O3 is highly exothermic and is used to join railway tracks. Identify metal X.Write the chemical equation for the reaction.
Answer: X is Al.
2Al + Fe2O3 → Al2O3 + 2Fe
Question: Write one example of each of
a. a metal which is so soft, that it can be cut with a knife and a non-metal which is the hardest known substance.
b. a metal and a non-metal which exist as liquid at room temperature.
Answer: a. Sodium is so soft that it can be easily cut with a knife.
b. Diamond is the allotrope of carbon, which is the hardest known substance and a non-metal.
c. Mercury is a metal and Bromine is a nonmetal which are found in liquid state at room temperature.
Question: Which of the following listed metals can displace zinc from its salt solution? Give reason for your answer with a chemical equation:
Copper, Lead, Magnesium, Silver
Answer: Magnesium can displace zinc from zinc salt solution
because Mg is more reactive than zinc:
Mg(s) + ZnSO4(aq) → MgSO4(aq) + Zn(s)
Question: Why is sodium chloride soluble in water but not in kerosene?
Answer: Sodium chloride is the ionic compound and it form ions in water, therefore it is soluble in water whereas kerosene is a non-polar covalent compound, NaCl does not form ions in kerosene, therefore, it is insoluble in it.
Question: Mention the name of metals for the following:
a. Two metals which are alloyed with iron to make stainless steel.
b. Two metals which are used to make jewellery.
Answer: a. Nickel and chromium.
b. Gold and platinum
Question: A copper plate was dipped into a solution of silver nitrate. After sometime a black layer was observed on the surface of copper plate. State the reason for it and write chemical equation for the reaction involved.
Answer: Copper is more reactive than Ag, therefore it can
displace Ag from AgNO3 solution:
Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s)
Question: Give reason:
a. Carbonate and sulphide ores are usually converted into oxides during the process of extraction of metals.
b. Ionic compounds generally have high melting point.
c. Hydrogen is not a metal but has been assigned a place in the reactivity services of metals.
d. The galvanised iron article is protected against rusting even if the zinc layer is broken.
Answer: a. It is easier to reduce an oxide than sulphide and carbonate ores.
b. It is due to strong forces of attraction between positive and negative ions.
c. Hydrogen form H+ ions like metals, therefore it is included in the activity series.
d. Zinc is more reactive than iron, therefore it will get easily oxidised in preference to Fe and hence protect iron from rusting even if zinc coating is broken.
Question: A compound Z is formed by transfer of electrons from the metal X to a non-metal Y, Identify the type of bond formed in the compound. List three properties of compound formed by such type of bonds.
Answer: The bond formed is ionic bond:
a. The compound will have high melting and boiling point.
b. It will be soluble in water.
c. It will conduct electricity in molten state as well as in aqueous solution.
Question: a. Why do aluminium sheets not corrode easily?
b. Why is copper vessel corroded with a green coating in rainy season?
Answer: a. Aluminium sheets are covered with an oxide layer which makes it passive.
b. It is due to the formation of basic copper
carbonate, CuCO3.Cu(OH)2.
Question: Out of the two metals P and Q, P is less reactive than Q. Suggest an activity to arrange these metals in the order of decreasing reactivity. Support your answer with a suitable chemical equation.
Answer: Add Q in the salt solution of P. If Q is able to displace P’ from its salt solution, then it shows P is less reactive than Q
Q + PR → QR + P
Question: Define alloy. How an is alloy prepared?
Answer: Alloy is an homogeneous mixture of two or more metals. One of them can be a non-metal also. Alloys are made by melting two metals together and then cooling it.
Question: Give reason for the following:
a. School bells are made up of metals.
b. Electrical wires are made up of copper.
Answer: a. Metals are sonorous i.e., produce sound when struck with a hard substance.
b. Copper is a good conductor of electricity and is highly ductile.
Question: Give reasons for the following:
a. Platinum, gold and silver are used for making jewellery.
b. Sodium, potassium and lithium are stored under kerosene oil.
c. Aluminium is highly reactive metal but still used for making cooking utensils.
d. Carbonate and sulphide ores are usually converted into oxides during the process of extraction.
Answer: a. It is because they are lustrous metals.
b. These are highly reactive metals.
c. It forms an oxide layer on its surface which makes it passive.
d. It is easier to reduce a metal oxide than sulphides and carbonates.
Question: In one of the methods of rust prevention, iron is not coated with anything. Name that method and define it.
Answer: The method is alloy formation, by adding a suitable metal or a non-metal.
Alloy is. a homogeneous mixture of two or more metals. One of them can be a non- metal also. This process is called alloying.
Question: Using the electronic configuration, explain how magnesium atom combines with oxygen atom to form
magnesium oxide by transfer of electrons.
Answer:
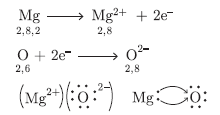
Question: Why hydrogen gas is not evolved when a metal reacts with nitric acid?
Answer: Nitric acid is an oxidising agent, therefore H2(g) is not evolved on reaction with metal. Dilute HNO3 mostly gets reduced to NO in this process.
Question: What happens to potassium and sodium if they are kept in open? Why are they 3 immersed in kerosene oil?
Answer: Potassium and Sodium if kept in open, can catch the presence of moisture.
They are immersed in kerosene so that they do not react with air and H2O present in the atmosphere.
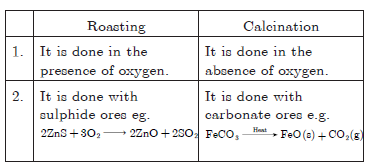
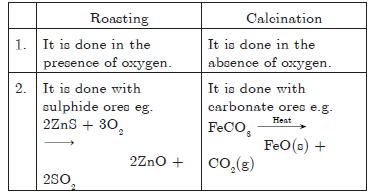
Question: Differentiate between roasting and calcination giving examples.
Answer:
Question: Name the ore of mercury. How mercury is extracted from its ore?
Answer: Cinnabar (HgS) is the ore of mercury.
Roasting:
HgS(s) + O2(g) → 2Hg(l) + SO2(g)
Cinnabar, on roasting gives Mercury and sulphur dioxide. Mercury can be purified by distillation process.
Question: Zinc does not evolve hydrogen gas on reacting with HNO3. Why?
Answer: Dilute HNO3 is an oxidising agent, therefore it does not liberate H2 with zinc. It oxidises N and liberates NO gas (Nitrogen monoxide).
Question: Give reason for the following:
a. Metals can be given different shapes according to our needs.
b. Hydrogen is not evolved when a metal reacts with nitric acid.
Answer: a. Metals are malleable, therefore they can be given different shapes.
b. It is because HNO3(dil.) is a good oxidising agent.
THREE MARKS QUESTIONS
Question: (a) What is reactivity series? How does the reactivity series of metals help in predicting the relative activity of various metals?
(b) Suggest different chemical processes used for obtaining a metal from its oxides of metals in the middle of the reactivity series and metals at the top of the reactivity series. Support your answer with one example each.
Answer: (a) The series in which metals are arranged in decreasing order of reactivity is called activity series of metals. The metal at the top is most reactive, followed by less reactive metal and so on. The metal at the bottom of activity series is least reactive.
(b) Metals at the top of activity series are obtained by electrolytic reduction e.g.,
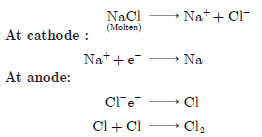
Metals in the middle of the reactivity series are obtained by reduction with A1 e.g.,
Cr2O3 + 2Al → Al2O3 + 2Cr
Question: Write one example of each of the following:
a. Most malleable and most ductile metal.
b. The best conductor of heat and poorest conductor of heat.
c. ‘A metal with highest melting point and a metal with lowest melting point.
Answer: a. Gold is most malleable and ductile.
b. Copper is the best conductor and lead is a poor conductor of heat copper.
c. Tungsten has highest melting point, mercury has lowest melting point.
Question: a. Write the steps involved in the extraction of pure metals in the middle of the activity series from their carbonate ores.
b. How is copper extracted from its sulphide ore? Explain the various steps supported by chemical equations. Draw a labelled diagram for the electrolytic refining of copper
Answer: a. Following steps are involved in the i. Hydraulic washing: The carbonate ores is washed with stream of water to remove earthly impurities.
ii. Calcination: Carbonate ores is heated strongly in the absence of air to form oxides e.g.,


Question: Aluminium oxide and zinc oxide react with both acids and bases to produce salt and water. What are these oxides called? Write chemical equations in each case.
Answer: These are called amphoteric oxides:
Al2O3 + 2NaOH → 2NaAlO2 + H2O
ZnO + 2NaOH → Na2ZnO2 + H2O
ZnO + 2HCl → ZnCl2 + H2O
Al2O3 + 6HCl → 2AlCl3 + 3H
Question: a. What is an alloy? How is it prepared? Give two examples of alloys.
b. Iron is not used in pure state. Give reason.
Answer: a. Alloy is a homogeneous mixture of two or more metals. One of them can be a non-metal also.
Alloys are made by melting two metals together and then cooling it. Example: Brass and Bronze
b. Iron gets rusted, therefore it is not used in pure state.
Question: Explain the following statements:
a. Most metal oxides are insoluble in water but some of these dissolve in water. What are these oxidesand their solution in water called?
b. At ordinary temperature the surface of metals such as magnesium, aluminium, zinc etc. is covered with a thin layer. What is the composition of this layer? State its importance.
c. Some alkali metals can be cut with knife.
Answer: a. These oxides are basic in nature and their solution are called alkalies.
b. The composition of this layer is metal oxide. It prevents the metal from corrosion.
c. Sodium can be cut with a knife because it is a very soft metal.
Question: A metal P when exposed to moist air for a longer period of time, loses its shining brown surface and attains a green coating; what has happened? Identify the metal, write the name and chemical formula of green coloured compound. List two ways to prevent this process.
Answer: It undergoes corrosion. The metal is copper.
Chemical formula of green coloured compound is CuCO3.Cu(OH)2
a. It can be prevented by coating of tin over iron.
b. It can also be prevented by coating it with paint.
Question: Name the constituent elements of alloys:
a. Brass,
b. Bronze,
c. Solder.
Mention one use of each alloy.
Answer: a. Brass is made up Cu and Zn. It is used for making decorative articles.
b. Bronze is made up of Cu and Sn. It is used for making statues, medals.
c. Solder it is an alloy of Pb and Sn. It is used for soldering purposes.
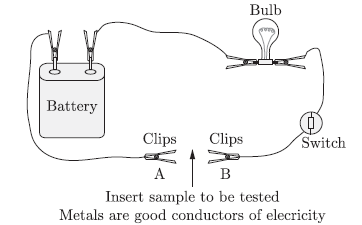
Question: Describe an activity to show that metals are good conductors of electricity.
Answer: Activity: To show metals are good conductors of electricity.
1. Take copper wire and insert two clips with a sample to be inserted between them, as shown in the diagram.
2. Set up the apparatus as shown in the diagram.
3. Plug the switch ON so that current starts flowing.
4. Record your observations.
5. Repeat the procedure with other metals.
Observation: The bulb glows with all metals.
Conclusion: Metals are good conductors of electricity.
Question: Name a metal in each case:
a. It displaces hydrogen gas on reaction with nitric acid.
b. It does react with any physical state of water.
c. It does not react with cold water or hot water, but reacts with steam.
Answer: a. Mg and Mn
b. Cu
c. Iron
Question: State three reasons for the following facts:
a. Sulphur is a non-metal.
b. Magnesium is a metal.
One of the reason must be supported with a chemical equation.
Answer: a. Sulphur is a non-metal because it reacts with O2 to form SO2 which is an
acidic oxide: S + O2 → SO2
Magnesium is a metal which reacts with O2 to form basic oxide:
2Mg + O2 → 2MgO
b. Sulphur is not malleable and ductile, magnesium is malleable and ductile.
c. Sulphur does not conduct electricity whereas magnesium conducts electricity.
Question: Give reasons only:
a. We can store copper sulphate solution in a silver vessel but not silver nitrate solution in copper vessel.
b. The reaction of zinc with dilute HNO3 does not produce H2 gas.
c. Food cans are coated with tin rather than zinc.
Answer: a. It is because copper is more reactive than silver, it will displace Ag from AgNO3 solution. Thus we can’t store AgNO3 in a copper container but CuSO4 can be stored in silver vessel as no reaction will take place because silver is less reactive.
b. Dilute HNO3 is an oxidising agent.
c. Tin is less reactive and less expensive than zinc.
Question: a. Write the electron-dot structure of calcium and sulphur.
b. Show the formation of CaS by transfer of electrons.
c. Name the ions present in the compound, CaS. Atomic number of Ca = 20, S = 16.
Answer:
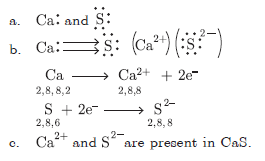
Question: A metal forms an oxide having formula M2O3. It dissolves both in dilute sulphuric acid as well as dilute sodium hydroxide solution. Identify the metal and write the equations for the reactions involved.
Answer:The metal is Al. Its oxide is Al2O3.
Al2O3 + 2NaOH → NaAlO2 + H2O
Al2O3 + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2O(l)
Question: Distinguish between the following:
a. Electrolytic reduction and electrolytic refining.
b. Mineral and ore.
c. Alloys and amalgams
Answer:

Question: What is cinnabar? How is metal extracted from cinnabar? Explain briefly.
Answer: Cinnabar (HgS) is an ore of mercury.
Roasting: HgS(s) + O2(g) → 2Hg(l) + SO2(g)
Cinnabar, on roasting gives Mercury and sulphur dioxide. Mercury can be purified by the process of distillation.
FIVE MARKS QUESTIONS
Question: a. Define activity series of metals. Arrange the metals: gold, copper, iron and magnesium in the order of their increasing reactivity.
b. What will you observe when:
(1) Some zinc pieces are placed in copper sulphate solution.
(2) Some silver pieces are placed into green coloured ferrous sulphate solution.
Answer: a. The series in which metals are arranged in decreasing order of reactivity is called activity series of metals. The metal at the top is most reactive, followed by less reactive metal and so on.The metal at the bottom is least reactive.
Au < Cu < Fe < Mg is the increasing order of reactivity.
b. (1) The solution will become colourless, reddish brown copper metal will get deposited:
Zn(s) + CuSO4(aq) → ZnSO4(aq) + Cu(s)
(2) No reaction will take place because Ag is less reactive than Fe.
Question: a. Write two differences between calcination and roasting.
b. ‘No reaction takes place when granules of a solid ‘A’ is mixed with a powder of solid ‘B’. However when the mixture is heated, a reaction starts with the evolution of much heat. Product ‘C’ of the reaction settles down as a liquid metal and a solid product ‘D’ keeps on floating over the liquid ‘C’.
This reaction is sometimes used for making metals for ready use in odd places.
a. Based on this information, make assumptions about metals ‘A’ and ‘B’ and corresponding deductions about ‘C’ and ‘D’ and write a balanced chemical equation for the reaction. Include in the chemical equation about the physical states of the reactants and products, need for heating for starting the reaction and the reaction being exothermic.
b. Name two types of chemical reactions to which this reaction can belong to.
Answer: a. Differences between calcination and roasting.
b. A is Al, B is Fe2O3, C is molten Iron, D is Al2O3
(Molten)
2Al(s) + Fe2O3(s) Heat→ Al2O3(l) + Fe(l)
A B D (Iron)
C
This reaction need heat to start but it is highly exothermic. This reaction belongs to the category of:
(1) Displacement reaction,
(2) Redox reaction.
Question: A metal (E) is stored under kerosene oil. When a small piece of it is left open in air, it catches fire. When the product formed is dissolved in water, it turns red litmus blue.
a. Name the metal (E).
b. Write the chemical equation for the reaction when it is exposed to air and when the product is dissolved in water.
c. Explain the process by which metal is obtained from its molten chloride.
Answer: a. E is sodium metal.
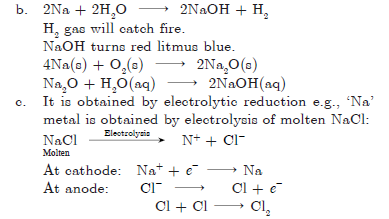
Question: Four metals A, B, C and D are added to the following aqueous solutions one by one. The observations made are tabulated below:
Answer the following questions based on the above observations:
a. Which is the most active metal and why?
b. What would be observed if B is added to copper
(II) sulphate solution and why?
c. Arrange the metals A, B, C and D in order of increasing reactivity.
d. Container of which metal can be used to store both zinc sulphate solution as well as silver nitrate solution?
e. Which of the above solutions can be easily stored in a container made up of any of these metals?
Answer: a. B is the most reactive because it displaces Fe. It
will displaces Fe. It will displace Cu as well as Ag also.
b. B will displace copper from CuSO4 solution because it is more reactive than Cu.
c. D is least reactive and D<C<A<B is the increasing order of reactivity.
d. Container of metal D can be used to store both ZnSO4 and AgNO3.
e. Zinc sulphate solution can be stored in container made up any of the metals A, B, C, D because none of them could displace zinc from zinc sulphate as these are less reactive than zinc.

